Cytochrome CYP2B6 Gene Polymorphism and Urinary Adverse Effects in Pakistani Breast Cancer Patients taking Cyclophosphamide
DOI:
https://doi.org/10.55627/pmc.002.01.0052Keywords:
Breast cancer, cyclophosphamide, renal toxicity, pharmacogenetics, CYP2B6, precision medicineAbstract
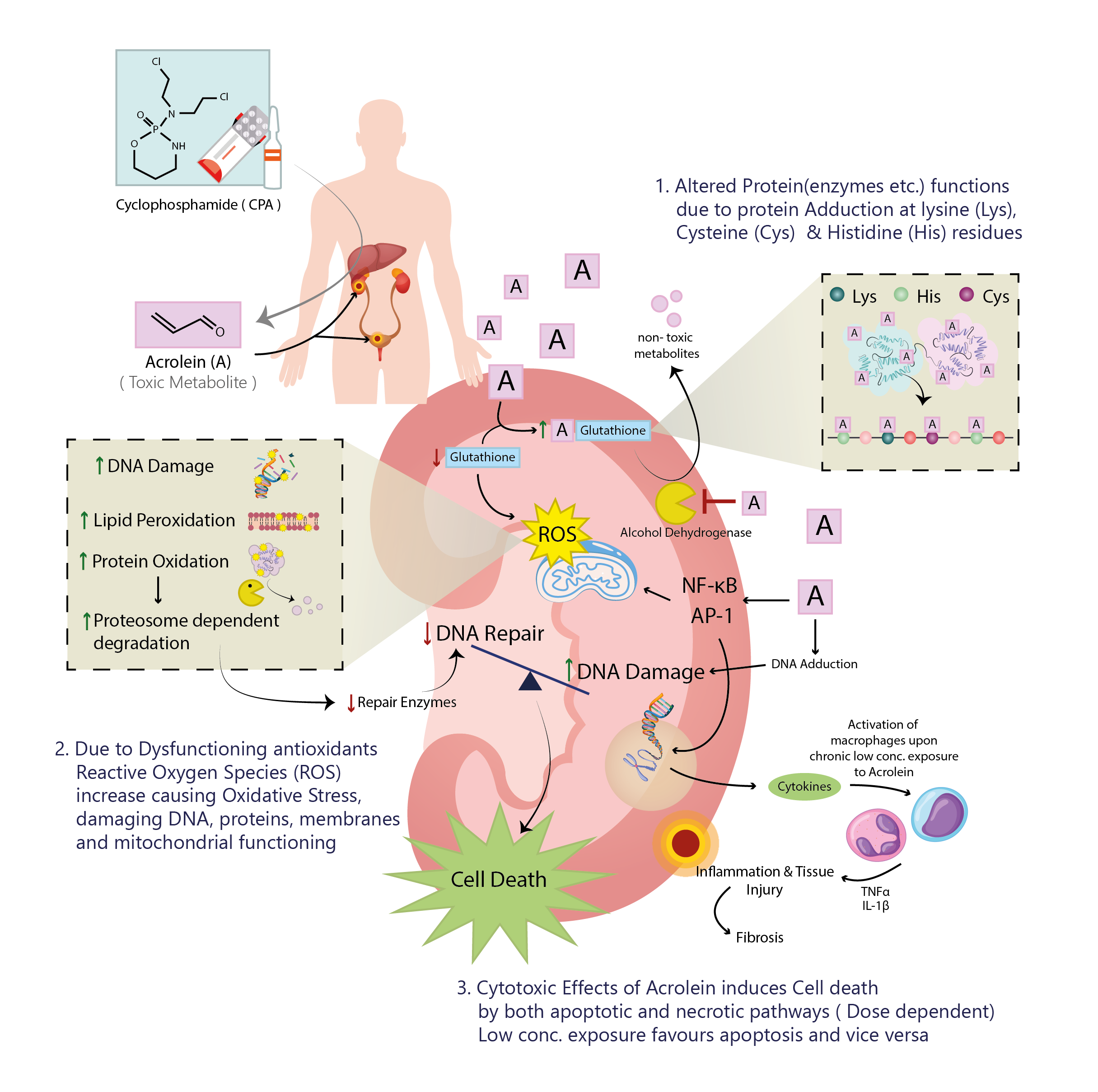
The use of cyclophosphamide is associated with renal and urinary adverse effects in some patients. Whether CYP2B6 gene polymorphism has anything to do with it is not very well known in the context of the Pakistani population. In this investigation, we studied the possible association between the single nucleotide polymorphism in cytochrome P450 2B6 and urinary adverse effects in a Pakistani cohort of breast cancer patients receiving cyclophosphamide as a part of therapy. In this cohort study, gene polymorphism of the CYP2B6 gene (CYP2B6 *6) of 51 histologically verified breast cancer patients were performed by using PCR-based restriction fragment length polymorphism. Our results show that among the patients with wild type (GG) genotype, about 78 % of patients did not experience any renal adverse effects while about 22% did. In patients heterozygous for the polymorphic genotype (GT), more than 81% did not complain of any renal adverse effects while only 18% showed renal toxicity. The group with TT genotype showed the highest fraction of patients experiencing renal adverse effects (25%). However, Chi-squared analysis did not reveal any association with renal adverse effects and with GT or TT genotype. Even when the analysis was repeated with merging genotypes GT and TT, there was no statistically significant difference between the wild type and GT+TT group. This study concludes that CYP2B6*6 is not associated with renal adverse effects in Pakistani breast cancer patients who are taking cyclophosphamide.
References
Published
Issue
Section
License
Copyright (c) 2022 Precision Medicine Communications

This work is licensed under a Creative Commons Attribution 4.0 International License.






