Assessment of Acute and Chronic Toxicity of the Active Fraction of Lavandula stoechas in Albino Mice: A Comprehensive Study
DOI:
https://doi.org/10.55627/ppc.004.002.0645Keywords:
L. stoechas, Acute Toxicity, Chronic Toxicity, AST, ALT, HematologyAbstract
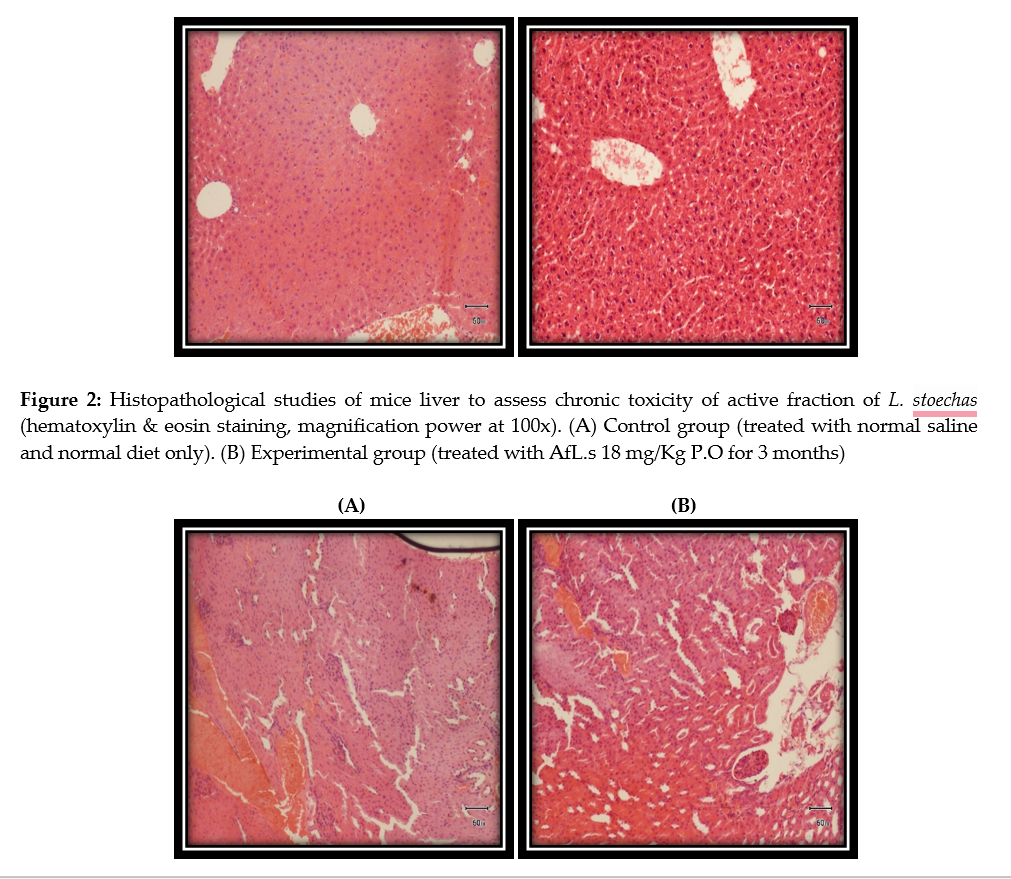
This study builds upon prior research where the principal components, phenethylamine and α-tocopherol, of Lavandula stoechas (L. stoechas) were identified as responsible for its nootropic potential. The current investigation aimed to determine the acute and chronic toxicity profiles of the active fraction of L. stoechas (AfL.s) in albino mice (ethical approval vide letter no; GCU-AEC-682/24). Acute toxicity assessment involved oral administration of AfL.s at doses of 350 mg/Kg and 300 mg/Kg, with toxicity signs monitored for 72 hours. For chronic toxicity evaluation, two groups of mice were administered either 10 ml/kg P.O. of normal saline or 18 mg/Kg P.O. of AfL.s daily for three consecutive months. At the end of the 90-day period, blood sampling was done via cardiac puncture to perform hematological and biochemical analyses. Additionally, the brain and liver were harvested for histopathological examination. The acute toxicity study revealed that AfL.s induced hyperexcitation, convulsions, ataxia, muscle spasms, and blanching in mice. Chronic toxicity evaluation indicated that a dose of 18 mg/Kg P.O. of AfL.s led to non-significant (P ≥ 0.05) alterations in hematological parameters; RBC count, platelet count, hemoglobin and serum biochemistry; cholesterol, creatinine, glucose, AST (aspartate transaminase), ALT (alanine aminotransferase), and TB (total bilirubin). However, WBC count, hematocrit, and alkaline phosphatase (ALP) levels exhibited slight increases, accompanied by a significant (P ≤ 0.05) reduction in body weight in the treated mice. Histopathological assessments did not reveal any signs of toxicity. Consequently, AfL.s at an oral dose of 18 mg/Kg P.O. is deemed safe for long-term use in mice.
References
Downloads
Published
Issue
Section
License
Copyright (c) 2024 Dr. Aamir Mushtaq, Dr. Tanzeela Rehman, Dr. Hamid Saeed , Dr. Sairah Hafeez , Dr. Amjed Hussain, Dr. Qurat Ul Ain, Dr. Umar Farooq Gohar

This work is licensed under a Creative Commons Attribution 4.0 International License.







