Cost of Pharmacogenetic-Guided Therapy in Type-2 Diabetes Mellitus
DOI:
https://doi.org/10.55627/pmc.001.01.0071Abstract
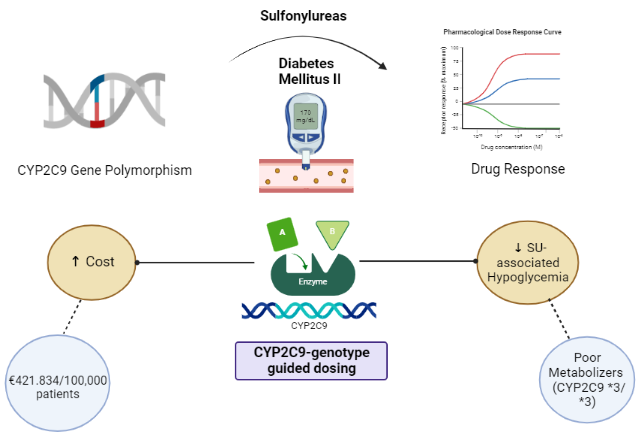
Connections between drug response and genetic variations may serve to optimize patient care. Drug response in type 2 diabetes mellitus (T2D) has been associated with a myriad of genetic polymorphisms. One pharmacological drug class commonly used for T2D management is Sulfonylureas. Incidences of severe hypoglycemia have been reported in patients that possess the CYP2C9*3/*3 variant of the enzyme in patients with co-administered glimepiride. Fokoun and colleagues investigated CYP2C9 genotype data to find out if it can assist in the management of sulfonylureas treatment regimens in patients that are about to start glimepiride. They also studied the possible impact on clinical and economic outcomes as well as the factors that can influence the cost-effectiveness of this approach. By adopting the perspective of the French national health insurance system, analysis was carried out over the course of 1 year using a decision tree. They found that €421 834, was the final cost for preventing a hypoglycemic episode per 100,000 individuals of which the genotyping cots proved to be the most notable factor in the incremental cost-effective ratio. Their study concluded that CYP2C9 genotyping guiding for glimepiride dosing yielded modest improvements in the frequency of hypoglycemic events but at a relatively higher cost. However, further studies are indispensable to determine whether this cost can be reduced further. Pharmacogenomics J. 2021 Oct;21(5):559-565.
Downloads
Published
Issue
Section
License
Copyright (c) 2021 Precision Medicine Communications

This work is licensed under a Creative Commons Attribution 4.0 International License.






