Genetic Variation in CYP2D6 and Adverse Effects in Cardiovascular Patients on Metoprolol Therapy
DOI:
https://doi.org/10.55627/pmc.004.001.0776Keywords:
CYP2D6, metoprolol, genetic variants, adverse effects, single nucleotide polymorphismAbstract
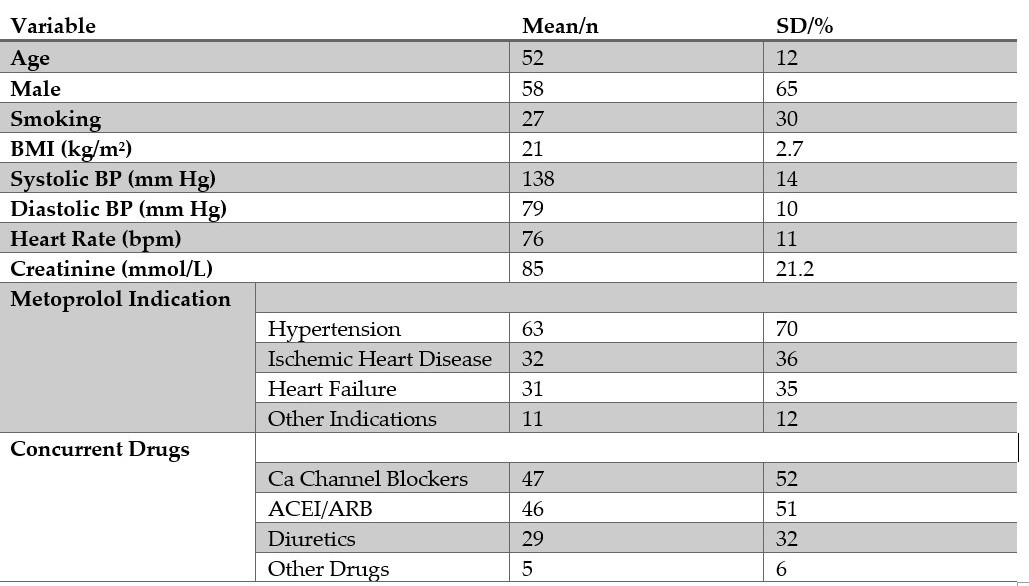
The CYP2D6 genetic variation seems to be important for several clinically used drugs including metoprolol. The frequency of adverse effects of metoprolol may vary depending upon the genotype of CYP2D6. However, how CYP2D6 genetic variation affects metoprolol's adverse effects is not well understood in the Pakistani population. In this study, we prospectively included Pakistani patients who started metoprolol treatment for cardiovascular indications. Decisions on inclusion and exclusion were made without awareness of the CYP2D6 genotype. Before intake of the first metoprolol dose, detailed medical history, baseline clinical characteristics, and laboratory indices were obtained. For CYP2D6 polymorphism examination, 5 ml of venous blood was collected and stored in a −80°C refrigerator until analysis. This study was approved by the Ethics Board of the Kohat University of Science & Technology and was conducted in line with the ethical guidelines of the 1975 Declaration of Helsinki. Our results showed that all the patients were wild type for this CYP2D6*6 (rs5030655) and no variant allele was found. For CYP2D6*4(rs1065852), 60 patients were wild type, 27 were heterozygous and only 3 patients were homozygous mutants for this single nucleotide polymorphism. Dizziness, bradycardia, and hypotension were more frequently observed adverse effects than lightheadedness and syncope. The frequency of adverse effects was hypotension (20), bradycardia (21), lightheadedness (16), dizziness (45), and syncope (13). The distribution of adverse effects in different genotypes of CYP2D6*4(rs1065852) reveals that this genetic variation is not associated with the adverse effects experienced by metoprolol users in a Pakistani cohort with cardiovascular indications.
References
Downloads
Published
Issue
Section
License
Copyright (c) 2024 Mehwish Rafique, Ayesha Nasir, Nida Saleem

This work is licensed under a Creative Commons Attribution 4.0 International License.






